
Research into Injury Data and E-scooter Risk
a study in collaboration with the Road Safety Authority (RSA)


PROJECT LEAD: John Legge & John Cronin
ITERN LEAD: Jeff Mulcaire, James Foley & Marcus Jee
RSA Leads: Stefania Costello , Velma Burns , Sharon Heffernan
Collaborators: Scroll to bottom of page
STUDY OUTLINE:
BACKGROUND:
E-scooter use and popularity has grown rapidly in recent years. This novel method for personal mobility comes with an increased burden of injury for road users. A pilot study at St Vincent’s University Hospital (SVUH) found a year-on-year increased presentation of e-scooter related injuries. This was a retrospective observational study over a 6-year period. In this study 326 patient presentations were assessed. 57% (n=188) were male and the median age was 32 years (range 14-87 years). While injury pattern varied, the majority of injuries were found on the upper limb, the face and head. The majority of patients (n=170) were discharged from the ED without any follow up required. The Road Safety Authority (RSA) has access to data from the National Trauma Audit, however patients who were managed and then discharged from the ED would not be included in this data set. For this reason the RSA approached the Irish Trainee Emergency Research Network (ITERN) with the view to collaborating on this study to improve their understanding of e-scooter related injuries.
Based on the study at SVUH, it is likely that large numbers of patients are presenting to other urban EDs as a result of e-scooter use. Although only a recent addition to our transport choices it is likely that e-scooter use is having a significant impact on presentations to the ED as a result of increased attendance. This study will review data retrospectively from a number of selected urban area EDs. New legislation was enacted in May 2024 regarding e-scooter use. This study should be able to assess the impact, if any as a result of this new legislation.
STUDY:
Service evaluation question
What is the prevalence and pattern of injuries associated with e-scooter use in Ireland and how does this impact on ED resources.
Study Design
This study is a retrospective review for the previous 3 years (January 1st 2021 to December 31st 2024 inclusive) and will take place across multiple urban area EDs. This will provide vital data on the impact that e-scooters are having on EDs across the country.
Primary Aim:
- Identify the prevalence of injuries associated with e-scooter use across all urban areas in Ireland
Secondary Aims:
- Identify if there was any impact from the enactment of legislation around e-scooter use
- Identify injury pattern of e scooter uses
- Patient disposition (discharged, admitted, transferred, etc)
- Patient length of stay if admitted
- Identify regional variation in e-scooter related presentations
- Quantify pedestrians, cyclists and other road users injured as a result of e-scooter use
- Assess for any regional variation in e-scooter presentations and injuries
- Quantify helmet use
- Quantify drug and alcohol use
DATA:
Inclusion Criteria
Patients presenting to the ED a result of an injury sustained from e-scooter use (e-scooter user, pedestrian, cyclist or other road user)
RESULTS: The results will be published in a relevant journal accessible to those who work in emergency medicine. The results will also be presented at national meetings, such as IAEM
AUTHORSHIP & COLLABORATIONS:
The main Authors will be of John Legge and his team. All who participate under the flag of ITERN will be given the collaborator status in the publication. These names will be indexed when the publication is finalised.
This study is being conducted in partnership with the RSA. This study will be published in a relevant Emergency Medicine Journal as well as the RSA website.
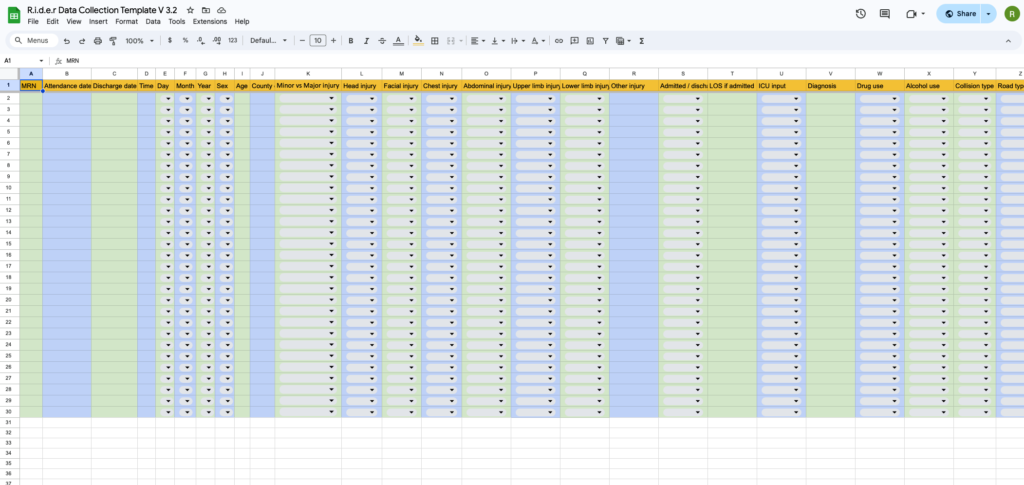
Site leads will be nominated at each participating ED. They will undergo training on how to conduct the retrospective study by identifying patients from the clinical records. A standardised data entry form will be generated which the site leads will populate with relevant data. This data will then be uploaded to the encrypted data storage software REDCap with patient identifiers removed. Data will be collected retrospectively covering a 3-year period. Patients will be identified from free word searches of triage and clinical notes.
TIMELINE: This study is targeted to commence in 2025, once a site lead and local ethical approval has been granted at each site. The retrospective data collection shall take place over 1-month period which should be adequate time for the site lead to collect the data but also allows extra time if a site experiences delays with site lead recruitment or ethics approval.
Data will be collected anonymously and up-loaded to REDCap. This platform is compliant with Good Clinical Practice and is regularly used to collect patient data for medical studies.
Who do I contact if I would like to take part in this study?
iterncommittee@gmail.com
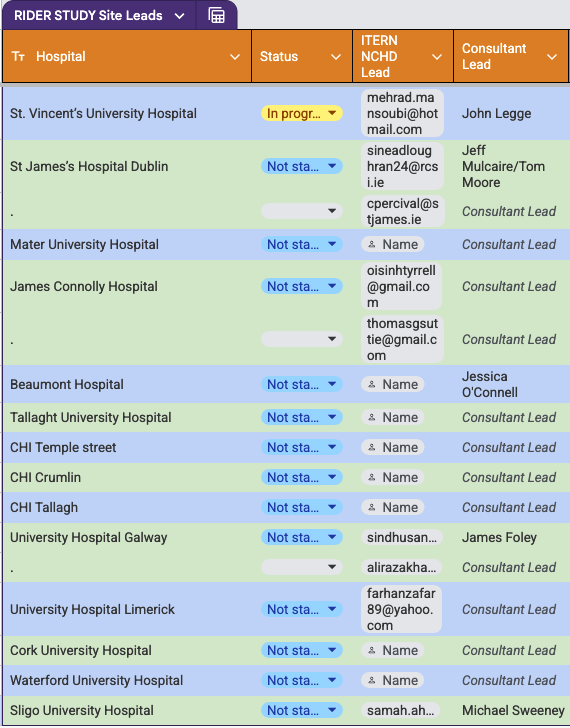
Collaborators (current) : Recruitment has not been finalised as of moment
If I am already involved as a site lead for data. what do I do?
IF you are already involved in this study, You will be invited formally via email shortly.
A study pack will be sent out containing all paperwork (incl. background, study design etc) to send to your local ethical committee and all of the steps/forms to ease collection of the data & a introductory zoom meeting will be planned.
If you did not receive the study packs you can use these links to access the study packs:
Download the study pack here + other essential documents
A link to a discord/whatsapp group will be sent to you and your name will appear in a table here on this page for confirmation when updated.